1.3. Synthesis of elements
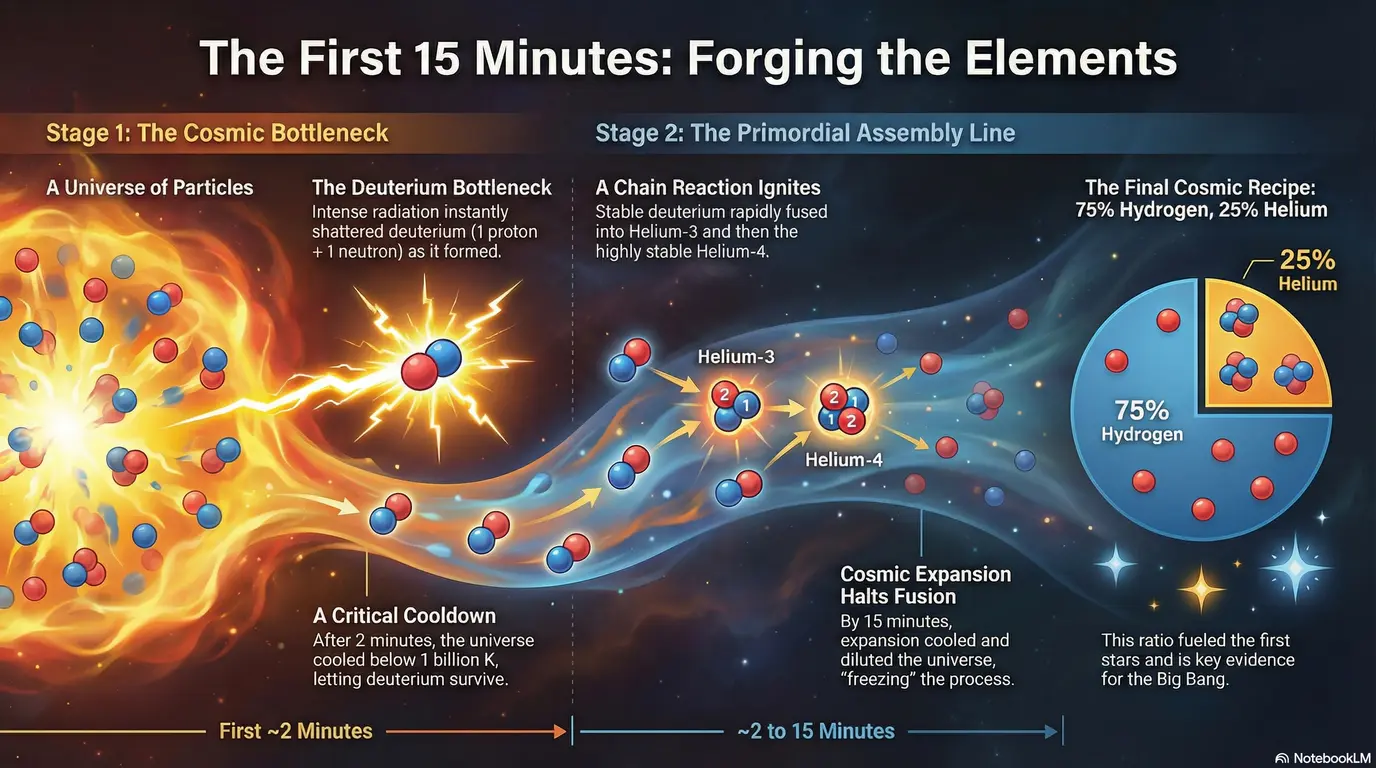
Immediately after the Big Bang, the universe was a blazing furnace of energy where temperatures were too extreme for even simple atoms to survive. Within microseconds, the cosmos cooled enough for fundamental quarks to combine into protons (made of two “up” and one “down” quark) and neutrons (made of one “up” and two “down” quarks). However, the path to building heavier elements was blocked by the “deuterium bottleneck.” For several minutes, intense radiation acted like a cosmic hammer, instantly shattering deuterium—a “heavy” hydrogen isotope made of one proton and one neutron—as soon as it formed. This fragile stepping stone was vital for cosmic evolution but could not endure the heat until the temperature fell below 1 billion Kelvin. Roughly two minutes after the start of time, the radiation finally eased, allowing deuterium to survive and triggering the next stage of creation.
Once this bottleneck was passed, a rapid and spectacular chain of primordial nucleosynthesis began, driven by a delicate tug-of-war between the fundamental forces of nature. Initially, the electromagnetic force acted as a barrier, using its positive charge to repel protons from one another. However, the immense thermal energy of the early universe provided these particles with enough speed to crash through that resistance and enter the tiny, powerful reach of the strong nuclear force, which acts like cosmic glue to bind particles together. In this brief window, deuterium nuclei quickly captured available protons to transform into Helium-3 (two protons, one neutron). These then swept up free neutrons—which were otherwise unstable and decaying due to the weak nuclear force—to create the incredibly stable Helium-4 nucleus, also known as an alpha particle. This burst of cosmic alchemy was so efficient that it locked almost all available neutrons into these sturdy helium structures before they had a chance to disappear.
This grand assembly line was extraordinarily brief, lasting only until the universe was about 15 minutes old. As the cosmos expanded at a staggering rate, its density and temperature plummeted below the levels required to fuse helium into more complex elements like carbon or oxygen. This rapid expansion effectively “froze” the chemical makeup of the early universe, leaving it with a simple but vital composition: roughly 75 percent hydrogen and 25 percent helium. If the expansion had been any slower, the universe might have fused all its material into iron—the most stable but inert element—resulting in a stagnant and lifeless cosmos. Instead, by stopping when it did, the universe preserved the vast reserves of hydrogen fuel that would eventually ignite the very first stars. Today, the fact that we still observe this precise 25 percent helium abundance and the presence of deuterium stands as one of the most powerful and undeniable pillars of evidence for the Big Bang.
