Table of Contents
Light
Light is a form of electromagnetic radiation consisting of periodically varying electric and magnetic fields that travel through a vacuum at a characteristic constant speed of approximately 299,792,458 m s$^{-1}$. While the term is often used to describe only the portion of the spectrum visible to the human eye, in a broader physical sense, light encompasses the entire electromagnetic spectrum, ranging from gamma rays and x-rays at the shortest wavelengths, through ultraviolet, visible, and infrared radiation, to microwaves and radio waves at the longest wavelengths. This radiation exhibits wave-particle duality, meaning it can be described both as a wave motion characterized by frequency ($\nu$) and wavelength ($\lambda$), and as a stream of discrete packets of energy known as photons ($\gamma$). The energy of these photons is inversely proportional to the wavelength; therefore, short-wavelength radiation such as gamma rays possesses high energy, while long-wavelength radiation like radio waves possesses low energy.
1. Nature of light
The electromagnetic nature of light, established by James Clerk Maxwell, implies that its propagation speed is governed by the fundamental electric and magnetic properties of the medium through which it travels. The equation
$$ c= \sqrt{\frac{1}{\epsilon\mu}} $$
expresses the speed in terms of the medium’s electric permittivity $\epsilon$ and magnetic permeability $\mu$, which relate the electric displacement and magnetic induction to the electric and magnetic fields, respectively. In the vacuum of free space, these physical constants are denoted as $\epsilon_0$ (the permittivity of free space) and $\mu_0$ (the permeability of free space). When electromagnetic radiation passes through a material medium rather than a vacuum, its velocity is reduced relative to $c$ by the index of refraction $n$, which is a function of the medium’s electromagnetic properties.

Light propagates through a vacuum as a transverse wave motion consisting of periodically varying electric and magnetic disturbances. These fields oscillate in a specific geometric relationship: the electric field vector ($\vec{E}$; $x$ axis) and magnetic field vector ($\vec{B}$; $y$ axis) are oriented transverse (perpendicular) to one another and also perpendicular to the direction in which the wave is propagating ($z$ axis).
The transport of energy associated with this wave motion is described by the Poynting vector ($\vec{S}$), a concept introduced by John Henry Poynting in 1884. This vector describes the rate at which energy is carried by the electromagnetic radiation. In the context of magnetohydrodynamics and electrodynamics, the energy conservation equations define $\vec{S}$ based on the cross product of the electric and magnetic fields, expressed as:
$$ \vec{S} = \frac{1}{\mu_0} \vec{E} \times \vec{B} $$
a vector that points in the direction of propagation, which corresponds to the direction of energy flow. The total energy density ($W$) of the wave is composed of contributions from both the electric and magnetic components. In a linear wave mode, the energy density of the thermal noise level is defined as the sum of the electric and magnetic energy densities as
$$ W_0 = \frac{1}{2}\epsilon_0 |E_0|^2 + \frac{1}{2\mu_0} |B_0|^2. $$
While both fields are required for propagation and carry energy, the electric field is conventionally used to define the polarization state of the radiation (e.g., linear, circular, or elliptical).
2. Speed of light
The speed of light, conventionally denoted by the symbol $c$, is a fundamental physical constant representing the speed at which electromagnetic radiation propagates through a vacuum. This characteristic speed applies to the entire electromagnetic spectrum, from radio waves to gamma rays, and links the wave-like properties of radiation through the fundamental relationship between frequency and wavelength. The relation between the frequency (the number of wave crests passing a point per second) and the wavelength (the distance between successive crests) is governed by the equation
$$ c=\nu \lambda $$
which implies that frequency and wavelength are inversely proportional; therefore, radiation with a shorter wavelength, such as blue light, travels at the same speed as red light but oscillates at a higher frequency.
Historically, the fact that light has a finite speed was first demonstrated in 1675 by the Danish astronomer Ole Rømer, who observed that the intervals between eclipses of Jupiter’s moon Io varied depending on the Earth’s distance from Jupiter. While early estimates were rough, precise terrestrial measurements were later achieved in the 19th and 20th centuries by physicists such as Fizeau, Foucault, and Michelson, the latter of whom used rotating mirrors and vacuum tubes to refine the value to near its modern accepted figure.
In the context of Albert Einstein’s special theory of relativity, the speed of light takes on a deeper significance as a universal constant that is the same for all observers in inertial frames, regardless of their relative motion, and serves as the absolute speed limit for the propagation of energy and matter in the universe,.
3. Electromagnetic spectrum
The electromagnetic spectrum encompasses the complete continuum of electromagnetic radiation, extending from low-energy radio waves to high-energy gamma rays, as shown in the figure below. This progression is defined by the fundamental inverse relationship between frequency and wavelength: as the frequency increases (measured in Hertz, spanning from approximately $10^3$ Hz to $10^{23}$ Hz), the wavelength decreases (measured in meters, ranging from $10^4$ m to $10^{-14}$ m). The spectrum is categorized into distinct bands: Radio (low, mid, high frequency), Microwave, Infrared (far, mid and near), Visible light, Ultraviolet, X-ray, and Gamma-ray.
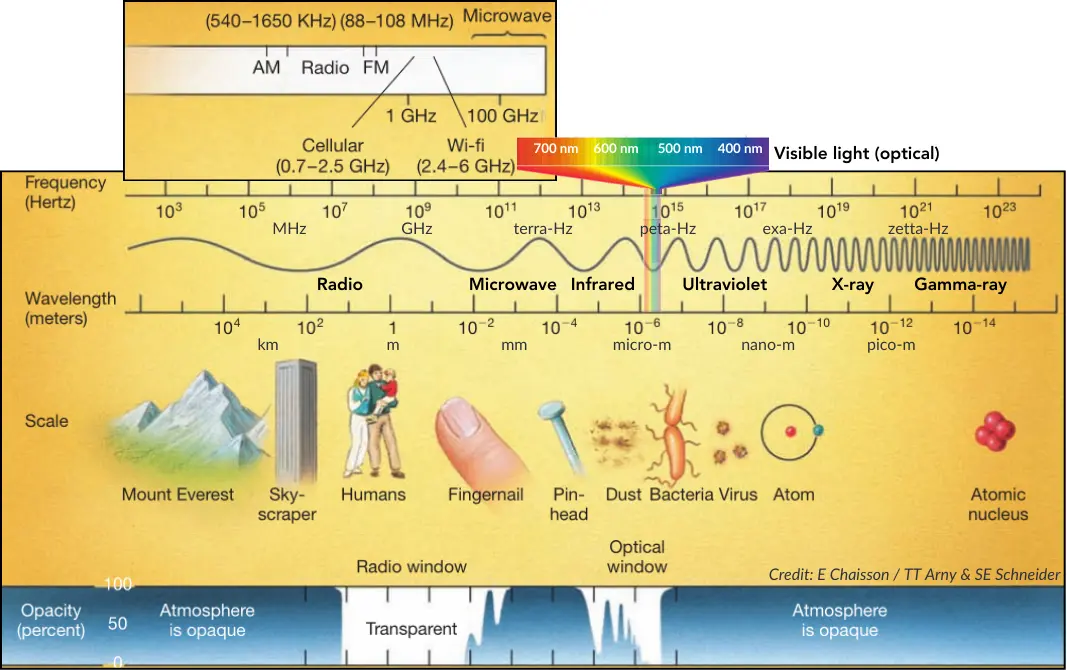
A specific section of the figure highlights the narrow band of visible light, expanded to show the optical spectrum. While boundaries can vary, the visible spectrum is typically defined as wavelengths between approximately $390$ nm (violet) and $750$ nm (red). Additionally, lower-frequency “communication” bands within the radio spectrum are noted, such as those for cellular networks and Wi-Fi.
To visualize the concept of wavelength, the scale of these waves is correlated with physical objects. Long radio waves are comparable to massive structures like skyscrapers or mountains. As the spectrum moves toward microwaves and infrared, wavelengths shrink to the scale of humans and small biological structures. Visible and ultraviolet waves correspond to the microscopic realm of dust and bacteria. Finally, the highest-energy X-rays and Gamma rays have wavelengths so infinitesimally small that they are comparable to the size of atoms and atomic nuclei.
The Earth’s atmosphere acts as a shield against most electromagnetic radiation, a property quantified as atmospheric opacity. Gamma rays, X-rays, and most ultraviolet and infrared radiation are blocked, meaning these signals cannot reach the ground. However, there are transparent areas: the “Optical window,” which allows visible light to pass, and the “Radio window,” which permits radio waves to reach the surface. Consequently, ground-based astronomy is largely restricted to optical and radio observations, while capturing other wavelengths, such as X-rays and Gamma rays, requires instruments placed above the atmosphere.
Everyday technologies exploit the unique properties of the electromagnetic spectrum across its various wavelengths. Low-frequency radio waves are utilized for standard AM/FM broadcasting and long-range communications. High-frequency radio and microwave bands support modern cellular networks (0.7–2.5 GHz), Wi-Fi (3.4–6 GHz), and radar systems used for navigation and tracking. Far-infrared radiation is primarily experienced as heat, playing a role in thermal sensing and management applications. Near-infrared technology is widely applied in night-vision devices for surveillance and in remote sensing to monitor the health of vegetation. Optical light is captured by cameras, which have evolved from simple pinholes to complex digital CCDs. Ultraviolet radiation is employed in “black lights” to induce fluorescence, a technique used for detecting forged documents. X-ray technology is fundamental to medical imaging and non-destructive testing of materials. Finally, gamma rays, resulting from radioactive decay, are associated with nuclear reactors and are monitored for radiation safety.
Unlocking the secrets of the cosmos requires specialized instruments for each slice of the spectrum. At the low-energy end, the Low Frequency Array (LOFAR) in Europe dominates the observation of low-frequency radio waves, while the Atacama Large Millimeter/submillimeter Array (ALMA) in Chile serves as the preeminent facility for high-frequency radio and microwave astronomy. Moving into the infrared, the James Webb Space Telescope (JWST) is the most powerful observatory ever built, optimized to capture near- and mid-infrared light from the earliest galaxies. For visible and ultraviolet light, the legendary Hubble Space Telescope (HST) remains the premier space-based instrument, complemented on the ground by the Very Large Telescope (VLT) in Chile. In the high-energy realm, Chandra X-ray Observatory provides the sharpest X-ray images of hot cosmic sources, while the Fermi Gamma-ray Space Telescope surveys the most energetic events in the universe, such as supermassive black hole jets and gamma-ray bursts.
