Table of Contents
This is an old revision of the document!
0.4. Light and Telescopes
Astronomers utilize telescopes as powerful time machines to investigate the seven ages of cosmic evolution, relying on the finite speed of light to view distant objects not as they are today, but as they existed many years ago when their radiation was first emitted. To probe the initial Particle Age, scientists employ radio telescopes to detect the Cosmic Microwave Background, the fossilized afterglow of the Big Bang. As the cosmos cooled into the Galactic Age, deep-field observations in visible and infrared light have revealed the chaotic assembly of the first galaxies, while multi-wavelength instruments allow astronomers to peer into the Stellar Age, penetrating dusty interstellar clouds to witness the birth of stars and the forging of heavy elements. This elemental enrichment sets the stage for the Planetary and Chemical Ages, where spectroscopes analyze starlight to identify complex molecules and planetary systems, providing the chemical foundation for the Biological Age. Finally, in the current Cultural Age, humanity turns these instruments outward to search for radio signals or laser pulses from other technological civilizations, to contextualize our own place in the extraordinary hierarchy of nature.
What is Light?
Light is a form of electromagnetic radiation (or waves) composed of rapidly fluctuating electric and magnetic fields that vibrate perpendicular to one another and to their direction of travel, moving through the vacuum of space at a constant, finite speed. This radiation arises whenever electrically charged particles, such as electrons, undergo acceleration or a change in motion; for instance, in a lightning bolt, accelerated charged particles release energy as visible light.
We characterize these waves by their wavelength—the distance between two consecutive wave crests—and their frequency, which is the number of crests that pass a specific point every second. These two properties share an inverse relationship, meaning that if you double the frequency, the wavelength is cut in half, because their combination must always equal the constant speed of light. Additionally, light behaves as discrete packets of energy known as photons, where the amount of energy carried is directly proportional to the frequency; consequently, radiation with a high frequency and short wavelength carries significantly more energy than radiation with a low frequency and long wavelength. The wavelength is measured in meters, frequency in hertz (Hz, cycles per second), and energy in joules.
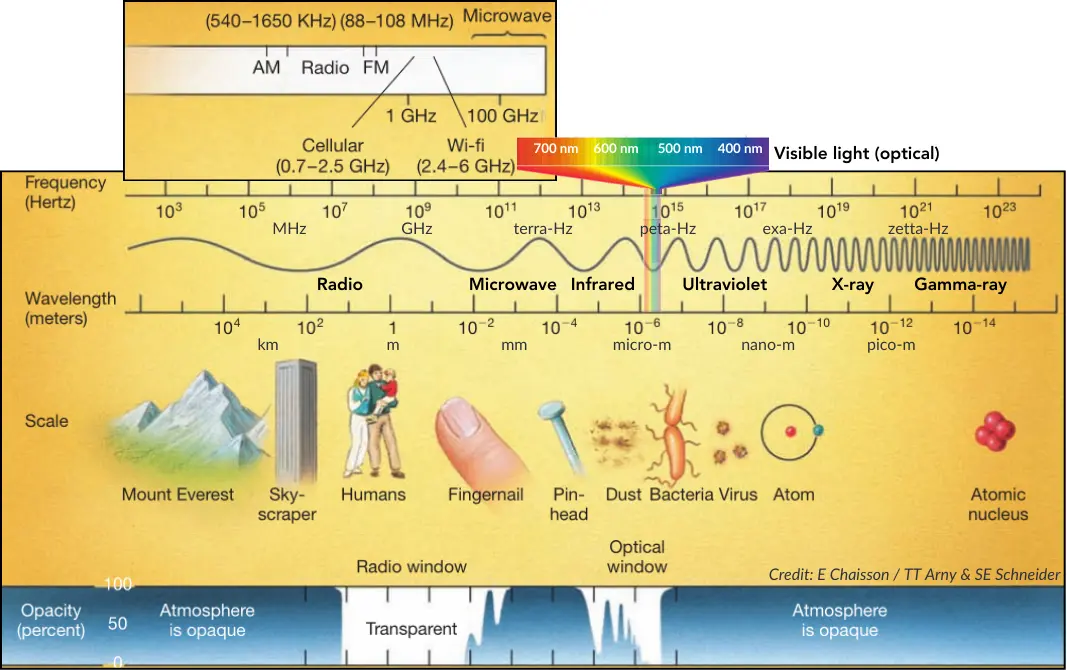
The electromagnetic spectrum begins with low-frequency radio waves, which possess the longest wavelengths—comparable to the scale of mountains—and the lowest energy, qualities that allow AM radio broadcasts to bounce off the atmosphere and travel over the horizon. As the frequency increases and wavelengths shorten, we encounter high-frequency radio waves, which carry slightly more energy and are utilized for FM radio and television signals that pass through the ionosphere rather than reflecting off it. Moving up the scale, microwaves feature even shorter wavelengths and higher frequencies, carrying sufficient energy to power our radar systems, Wi-Fi networks, and cellular communications. As energy intensifies further, we reach infrared radiation, which we physically experience as heat and utilize in technologies like remote controls and night-vision sensors that see through dark or dusty conditions.
This transitions into the narrow band of visible light, the only radiation possessing the specific frequency and energy required to trigger chemical reactions in human eyes, allowing us to perceive the colors of our world. Just beyond the violet end lies ultraviolet radiation, which has shorter wavelengths and carries enough energy to penetrate and damage living cells, a process we experience in daily life as suntans or sunburns. At even higher energies are X-rays, which have such high frequency and penetrating power that they can pass through soft tissue to reveal the shadows of our bones in medical imaging. Finally, the spectrum culminates with gamma rays, which possess the shortest wavelengths and the highest energy—billions of times that of visible light—and are associated with the intense nuclear reactions of radioactivity and nuclear explosions.
Earth’s atmosphere functions as a selective shield, effectively opaque to most electromagnetic radiation but permeable through specific “spectral windows” where the air is transparent,. The most significant openings are the optical window, allowing visible light to reach the surface, and the radio window, which permits the passage of radio waves with wavelengths ranging from approximately one centimeter to ten meters. Conversely, atmospheric gases absorb the majority of other wavelengths; the ozone layer completely blocks lethal ultraviolet, X-rays, and gamma rays, rendering these energetic bands observable only from space. Infrared radiation encounters a “partially transparent” atmosphere, where water vapor and carbon dioxide absorb many wavelengths, though some infrared light can still reach high, dry mountain observatories. Finally, the electrically conductive ionosphere reflects radio waves longer than about 10 meters, preventing them from penetrating to the ground.
How Telescopes Work
To explore the full breadth of the electromagnetic spectrum, astronomers utilize a diverse array of specialized instruments, starting with radio waves that are captured by massive ground-based dishes like the 100-meter Green Bank Telescope and the interferometric Very Large Array (VLA) to map cold interstellar gas and active galaxies. Moving to microwaves, space-based observatories such as COBE, WMAP, and Planck have been essential for mapping the cosmic microwave background radiation without atmospheric interference. To detect infrared radiation, which is often blocked by water vapor in Earth’s atmosphere, astronomers use space telescopes like Spitzer and Herschel, or high-altitude ground observatories like those on Mauna Kea, to peer through dust and study star formation. The visible spectrum is captured by both giant ground-based instruments like the Keck and Very Large Telescope (VLT) and the orbiting Hubble Space Telescope, which provides sharp images free from atmospheric blurring. Higher-energy ultraviolet light, largely absorbed by the ozone layer, is monitored by satellites such as GALEX and FUSE to study hot, young stars. X-rays from violent events like supernova remnants are focused using nested grazing-incidence mirrors on satellites like the Chandra X-ray Observatory and XMM-Newton. Finally, the highest-energy gamma rays are detected by missions like the Fermi Gamma-ray Space Telescope and Swift, which do not focus light but rather count individual high-energy photons emitted by cataclysmic events like gamma-ray bursts.
Every modern telescope system operates through three integrated stages to transform faint cosmic signals into viewable data. The process begins with a collector, for example, a large curved primary mirror, which functions as a “light bucket” to intercept incoming radiation and concentrate it into a focused beam at a specific point. Positioned at this focus is a detector, for example, a Charge-Coupled Device (CCD) containing millions of light-sensitive pixels, which converts the impacting photons into a corresponding buildup of electrical charge (electrons) proportional to the light’s intensity. Finally, these electrical signals are read out to a processor, where computers digitize the raw data, reduce background noise, and mathematically compensate for instrumental defects to reconstruct the electron counts into the detailed visual images used for analysis. This technology is not limited to high-end astronomy; the digital cameras found in smartphones and home video recorders function the same way, using these chips to capture everyday images electronically rather than on film.
Beyond capturing visual images, telescopes can act as analytical laboratories by inserting a spectrograph or spectrometer between the collector and the detector. In this configuration, the light gathered by the primary mirror is not focused directly into a picture but is first passed through a prism or grating to be dispersed into its component frequencies—creating a spectrum analogous to a rainbow—before the photons strike the CCD to be converted into electrons. The processor then reconstructs this data into a detailed graph of intensity versus wavelength rather than a 2D image. This spectral analysis is vital for astronomers because it reveals the unique “fingerprints” of matter; by studying these specific frequencies, scientists can identify the chemical composition, temperature, and velocity of celestial objects, effectively allowing them to decipher the physical nature of the universe from billions of light-years away.
