Table of Contents
Atmosphere
Stars, planets, and moons can all have atmospheres. Here we will mainly talk about the atmospheres of planets.
1. Atmospheric Pressure
To begin, we need an equation for atmospheric pressure, which can be derived using hydrostatic equilibrium. The relationship between pressure ($P$) and height ($z$) from the surface is:
$$ \frac{dP}{dz} = -\rho g $$
where $g$ is the gravitational acceleration and $\rho$ is the density of air. The equilibrium of the interior of a star or planet is also explained using a similar kind of equation. The relation between density and pressure can be written using the ideal gas law as:
$$ \frac{P \mu_a m_a}{\rho} = kT $$
where $k$ is the Boltzmann constant, $T$ is the air temperature, $\mu_a$ is the mean molecular weight (dimensionless), and $m_a$ is the atomic mass unit. Because in the equation $PV=NkT$, we can write $V/N=(M/\rho)/N= (M/N)/\rho = \mu_am_a/\rho$, where $M/N=\mu_a m_a$ is the average mass of an air particle in kilograms. From the two equations above we get:
$$ \frac{dP}{dz} = -\frac{P\mu_a m_a g}{kT} $$
Solving this gives us how pressure changes with height. The solution is easier if we assume $g$ and $T$ to be constant. For Earth’s atmosphere, this assumption isn’t too bad, because gravity hardly changes, and temperature changes much more slowly than pressure. The above differential equation can be solved by variable separation as follows:
$$ \int\frac{dP}{P} = -\frac{\mu_a m_ag}{kT} \int dz \Rightarrow \ln P = -\frac{\mu_a m_agz}{kT}+C $$
$$ \Rightarrow P = P_0 e^{-\mu_a m_a gz/(kT)}$$
where $P_0=e^C$ is the pressure at sea level ($z=0$), and $C$ is the integration constant. From the final equation it is evident that, at least for Earth, pressure decreases rapidly with height from the surface, because $P\propto e^{-z}$—that is, pressure decreases exponentially with altitude.
Important observatories like the Keck Observatory are located atop Hawaii’s Mauna Kea mountain; at 4 km altitude, the pressure is only 60% of sea level pressure. At the top of Everest, i.e., 9 km up, the pressure is just 30% of surface pressure. If pressure drops so drastically within just 9 km, it means that Earth’s atmosphere is very thin compared to its radius ($R_E$: 6,000 km). That’s why treating $g$ as constant was not a bad assumption; the acceleration doesn’t change much for just a 10–20 km change in height.
2. Mass of the Atmosphere
The mass $M$ of Earth’s atmosphere can be easily measured. The total gravitational force of the atmosphere at sea level is $F=Mg$, then the pressure $P_0=F/A=(M/A)g$, and therefore
$$ M = \frac{P_0A}{g} = \frac{4\pi R_E^2P_0}{g} $$
which comes out to approximately $5\times 10^{18}$ kg, or about one millionth of Earth’s total mass.
3. Earth’s Atmosphere

This plot shows how temperature and pressure change with altitude in Earth’s atmosphere. Pressure is given in millibars; 1 bar equals 100,000 pascals. At ground level, the pressure is about 1 bar, or roughly 1000 millibars. The temperature here is around 288 Kelvin, which keeps dropping up to the tropopause. Earth absorbs visible light from the Sun and emits infrared radiation; since it is itself a radiating source, temperature drops the higher you go from its surface. This is the reason for the temperature drop in the troposphere. All clouds and our weather systems exist in this region.
After the tropopause, temperature begins to rise again because in the stratosphere lies our protective ozone (O$_3$) layer. Because this molecule has three oxygen atoms instead of two, it absorbs ultraviolet light from the Sun, which raises its temperature, while shielding us below. This trend continues up to the stratopause, after which in the mesosphere temperature begins to drop again because ozone is absent. Temperature continues to drop up to the mesopause at around 90 km altitude, after which it starts rising again as sunlight directly affects the atmosphere in various ways in the thermosphere. The plot doesn’t show what lies above, but higher up at around 500 km is the exobase, from where the exosphere begins. In the exosphere, air density is extremely low, and the fast-moving molecules gradually escape into space.
Our atmosphere contains 78% nitrogen, 21% oxygen, and then trace amounts of argon, water vapor, and carbon dioxide. On Venus, 97% is CO$_2$, and on Mars 95% is CO$_2$. Saturn’s moon Titan has 90% to 97% nitrogen.
4. Atmospheres of Other Planets
The table below shows the amounts of various elements and compounds present in the atmospheres of three inner planets and Saturn’s moon Titan. Mercury is not included because Mercury has no significant atmosphere to speak of. All amounts are given in parts per million (ppm). On Earth, oxygen being 210,000 ppm means that for every one million particles in Earth’s atmosphere, 210,000 are oxygen.
| Molecule | Earth | Venus | Mars | Titan |
|---|---|---|---|---|
| Nitrogen (N₂) | 780000 | 35000 | 27000 | 900000–970000 |
| Oxygen (O₂) | 210000 | 0–20 | ||
| Argon (Ar) | 9000 | 70000 | 16000 | 48000 |
| Water vapor (H₂O) | <30000 | 50 | <100 | |
| Carbon dioxide (CO₂) | 345 | 970000 | 950000 | |
| Neon (Ne) | 18 | 7 | 2.5 | <0.01 |
| Ozone (O₃) | 10 | |||
| Helium (He) | 5 | 12 | ||
| Methane (CH₄) | 3 | 50000 | ||
| Krypton (Kr) | 1 | |||
| Carbon monoxide (CO) | 50 | 700 | 10 | |
| Nitric oxide (NO) | 3 | |||
| Sulfur dioxide (SO₂) | 60 | |||
| Hydrogen (H₂) | 10 | 0.002 | ||
| Acetylene (C₂H₂) | 2 | |||
| Ethane (C₂H₆) | 10 |
Earth’s atmosphere contains mostly nitrogen and oxygen, but the main component of the atmospheres of Venus and Mars is carbon dioxide. In terms of abundance, carbon dioxide ranks fifth in Earth’s atmosphere. Although Titan has a lot of nitrogen like Earth, it also contains organic compounds such as methane.
We know less about the atmospheres of the four outer planets. The Galileo probe entered Jupiter’s atmosphere, but was destroyed by extreme pressure shortly after entry, in 2003. The giant planets have very thick atmospheres, so even with distant imaging it is hard to get much detail about their interiors. Still, a lot has been learned from remote observations. Optical and ultraviolet light cannot penetrate their atmospheres, so more can be seen using infrared, submillimeter, and radio waves. The atmospheres of the outer four giants are mainly composed of hydrogen and helium. However, clouds of methane, water, ammonia, and hydrogen sulfide are also found, because the temperatures are low enough for these gases to condense; in Jupiter and Saturn, their abundance is about 1 percent.
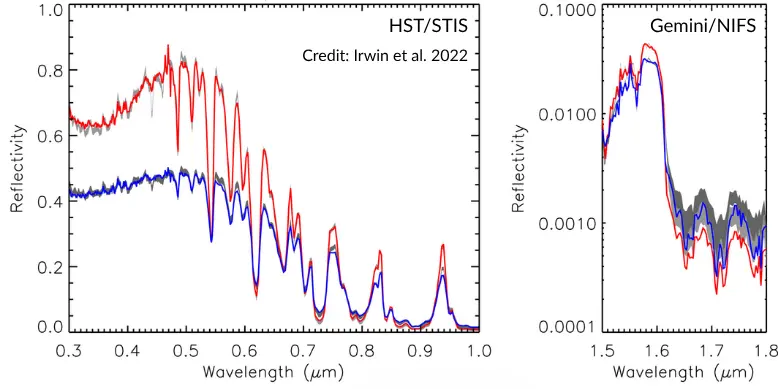
The atmospheres of Uranus and Neptune contain about 3 percent methane, and it is this gas that gives them their blue color. Above are spectra of the two planets — the left one observed using the STIS instrument on the Hubble telescope, and the right one using the NIFS instrument on the Gemini telescope at Mauna Kea. It can be seen that between 0.3 and 1.8 micrometers, both planets reflect blue light the most, and after 0.6 micrometers red light begins, whose reflectivity (i.e., albedo) is much lower for both planets. Also, between 0.3 and 0.6 micrometers, Neptune’s (red line) reflectivity is higher than Uranus’s (blue line).
Temperature variations in the atmospheres of giant planets have also been detected through spectroscopy. A simple explanation of how this works can be given as follows. Suppose continuum radiation is coming from a planet’s surface — meaning light is received at all frequencies within a certain range — and from various layers above the surface in the atmosphere come line radiations, meaning absorption or emission lines at specific frequencies. If the layer above is cooler than the layer below, there will be absorption in the upper layer; if the reverse is true, there will be emission. Observations have shown that in the atmospheres of giant planets, temperature decreases from the surface upwards, and the rate of decrease is similar to the adiabatic gradient in Earth’s troposphere (see clouds).
Titan is the only moon with a significant atmosphere. Just as water can exist in all three states — solid, liquid, and gas — on Earth, methane can exist in all three states on Titan. This means Titan has methane lakes and ice, and methane rain and snow fall from Titan’s skies. The Huygens probe, which landed on Titan’s surface, observed methane vapor rising from the surface, meaning that Titan has a methane cycle similar to Earth’s water cycle.
However, a good quality map of Titan’s surface was possible thanks to the radar on the Cassini satellite orbiting it. Cassini’s transmitter sent radio waves to Titan’s surface, part of which were reflected back, and Cassini’s receiver analyzed them to create a surface map. Through this kind of astrogeology, we now have a geological map of Titan, shown above. Cassini found more than seventy locations on Titan’s surface that reflected very little radio signal, just like seas or lakes on Earth. These methane (and ethane) reservoirs range in size from 3 to 70 km and are located towards the poles, especially in the northern polar region. As elsewhere in the Solar System, lakes are called lacus and seas are called maria. In the image, Kraken Mare near the north pole and Ontario Lacus near the south pole are shown in blue.
5. Birth of atmospheres
The atmospheres of the four inner planets are not primary, but secondary. If an atmosphere is formed during the process of a planet’s formation itself, it is called a primary atmosphere, and if it forms afterwards through other processes, it is secondary. One strong piece of evidence that Earth’s atmosphere is not primary is that, if it were, the abundances of various elements in it should have been like those in the primordial solar nebula, and thus similar to the overall averages found across the current Solar System. In reality, the proportions of elements in our atmosphere differ greatly from the Solar System average. In the Solar System, hydrogen and helium are the most abundant, but in our atmosphere nitrogen and oxygen dominate.
Still, those supporting a primary atmosphere might argue that in the beginning our atmosphere also had a lot of hydrogen and helium, but since they are the lightest elements and our planet’s mass is not very large, Earth’s gravity could not hold on to them, and they escaped into interplanetary space. This reasoning runs into trouble with neon. Neon is fifth in abundance in the Solar System, yet none of the four inner planets has much neon. Also, neon is not so light that the gravity of these planets could not retain it. If the atmospheres of Venus, Earth, and Mars were primary, then where did all the neon go? Since neon is an inert gas, it could not have reacted with other elements and gone into the ground. The only explanation can be that our atmosphere is not primary, but secondary.
This means that the atmospheres of the three rocky planets formed through two secondary processes: the release of gases from their interiors, and impacts with ice-rich planetesimals after their formation. Many planetesimals merged to form the planets, and many of them already contained rock and ice. So the materials needed to make an atmosphere were present inside the planet from its birth. Just as gases are released from volcanoes today, a similar process probably released large amounts of gas from Earth’s interior soon after its formation to create its atmosphere. This process is slow. By contrast, a collision with another ice-rich planetesimal could have created an atmosphere much more quickly.
An atmosphere can be reducing (hydrogen-based) or oxidizing (hydrogen-poor). Reducing air contains large amounts of methane, ammonia, water, and hydrogen sulfide. Oxidizing air contains carbon dioxide, nitrogen, and sulfur dioxide. The current atmospheres of rocky planets are mostly oxidizing, but whether they were so in the beginning is hard to say. One general truth, however, is that for an atmosphere to exist, a planet must be massive enough, which can be understood by writing the equation for the Maxwell–Boltzmann distribution for all the particles in the air:
$$ p(>v_{\text{esc}}) = \int_{v_{\text{esc}}}^{\infty} N \left( \frac{2}{\pi} \right)^{\frac{1}{2}} \left( \frac{m}{kT} \right)^{\frac{3}{2}} v^2 e^{-\frac{mv^2}{2kT}} \, dv $$
Here the tail of the distribution is shown, meaning $p(> v_{esc})$ is the fraction of particles whose speed is greater than the planet’s escape velocity, and which therefore cannot be held by the planet’s gravity. Here $N$ is the number density of the gas particles, and $m$ is the mass of each particle. The equation shows that increasing the particle mass or the object’s escape velocity means fewer particles can escape. Since hydrogen has very low mass, it has escaped more from the atmospheres of rocky planets, which is why even if their atmospheres were once more reducing, they are now more oxidizing. And the more massive the object, the higher the escape velocity needed to leave it. This is why smaller planets or moons have less ability to retain an atmosphere.
Although Earth has a lot of water, Venus has almost none. Perhaps Venus formed from planetesimals that did not contain much water. Or it could be that Venus’s atmosphere originally had a lot of water, but being closer to the Sun, ultraviolet light broke its water into hydrogen and oxygen, with the lighter hydrogen escaping. This process is called photodissociation. There are also many other processes by which air can be lost from an atmosphere. In the case of Mars, its atmosphere is being lost mainly because its mass is much smaller.
A molecule in an atmosphere, however, can also be lost not to space, but by entering the planet’s interior, as in Earth’s carbonate–silicate cycle. In this cycle, carbon dioxide is removed from the atmosphere and stored on the surface, and later returned to the atmosphere. First, carbon dioxide is washed down by rainwater to the ground, where it reacts with silicates in rocks to produce calcium and carbonate ions. These flow with river water into the ocean, where various organisms use them to build calcium carbonate shells. After death, these shells accumulate on the ocean floor. The carbon dioxide stored in this way is returned to the atmosphere through tectonic activity. The oceanic crust is subducted into Earth’s mantle, where it melts under high temperatures and reacts with silicates to produce carbon dioxide, which then escapes from Earth’s interior during volcanic eruptions and returns to the atmosphere. Because of this cycle, the amount of carbon dioxide in the atmosphere — and thus the temperature — has remained roughly constant over the past four and a half billion years, even though the Sun’s brightness has increased by 30% during this time.
